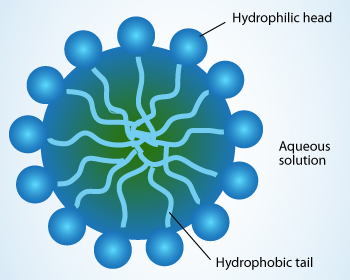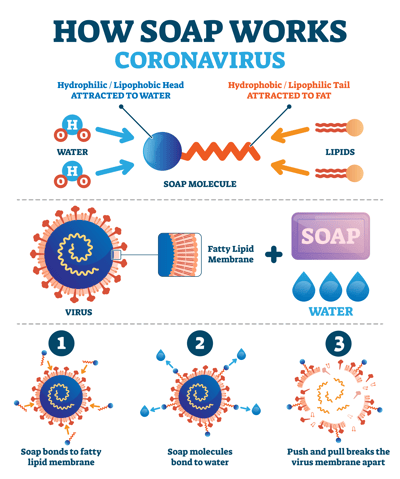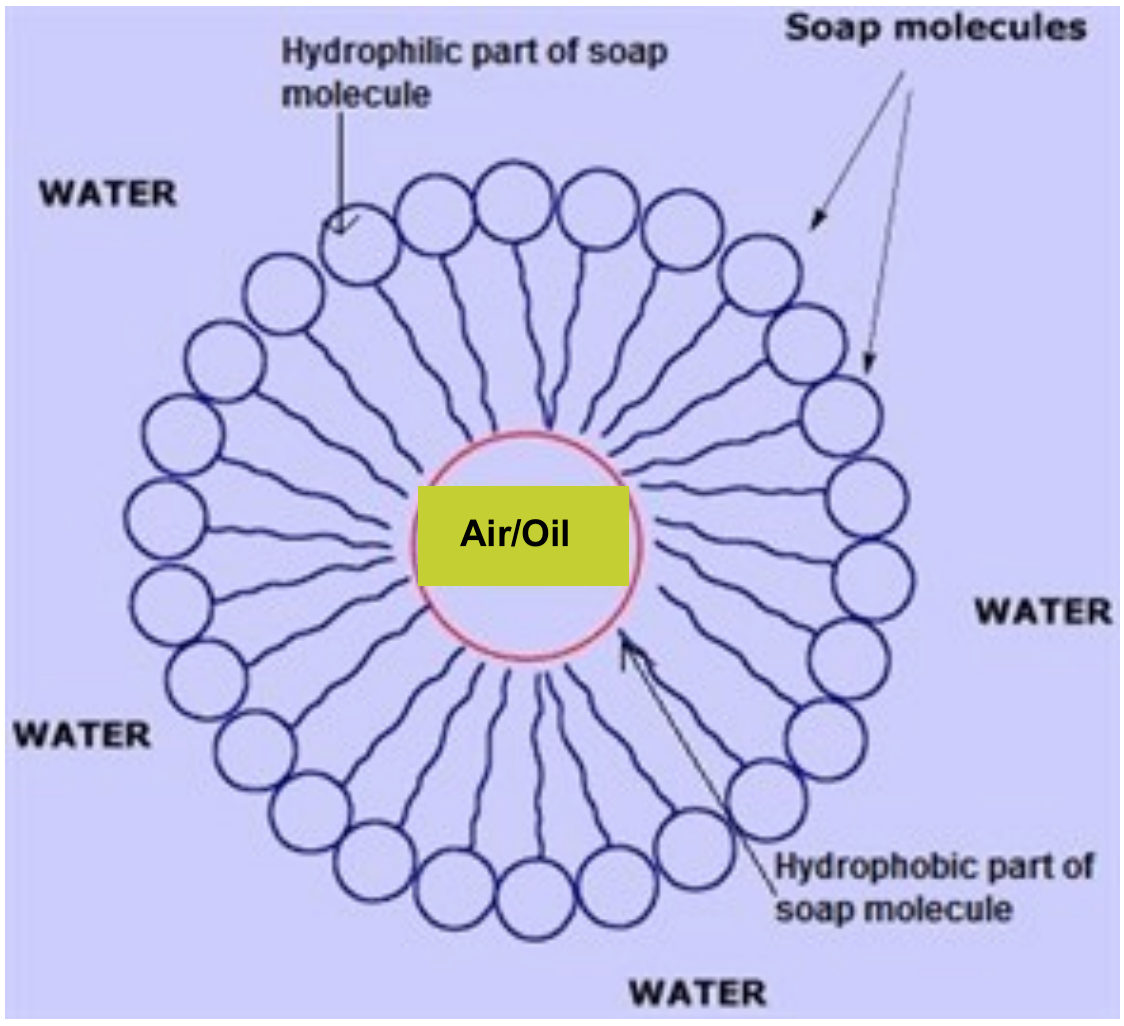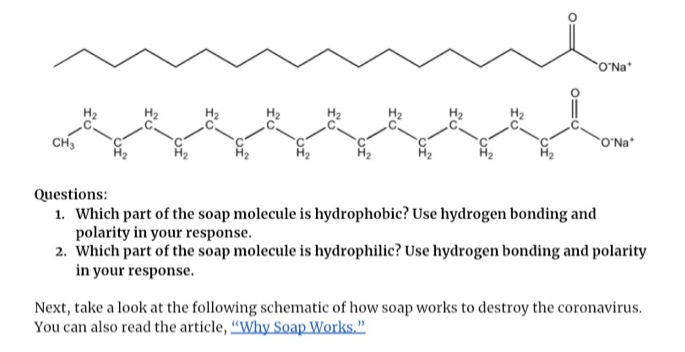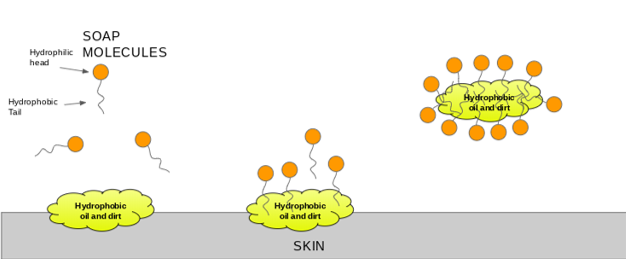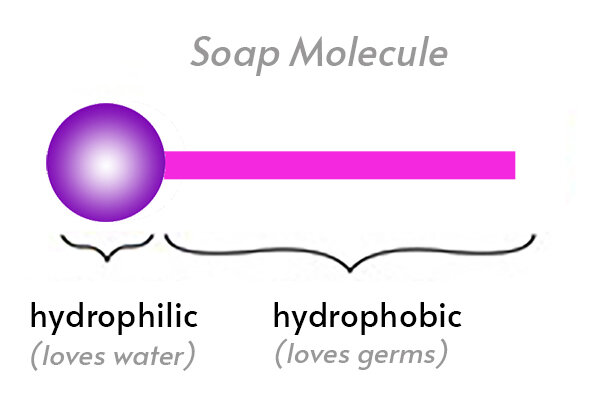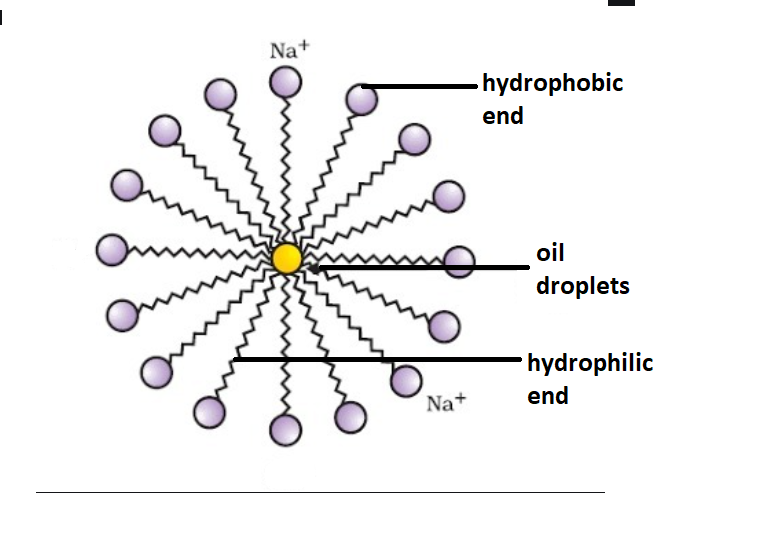
Which of the given statements is true?a.) The ionic end of soap dissolves in water while the carbon chain dissolves in oilb.) The ionic end of soap dissolves in oil while the
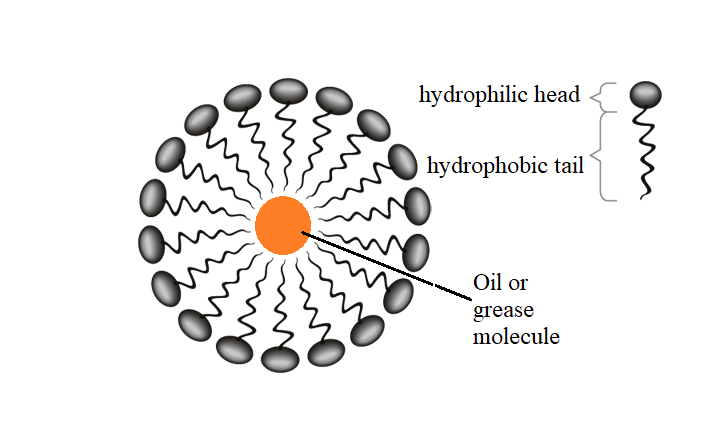
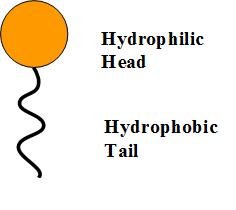
The soap molecule has a: A) Hydrophilic head and a hydrophobic tail B) Hydrophobic head and a hydrophilic tail C) Hydrophobic head and a hydrophobic tail D) Hydrophilic head and a hydrophilic

Question 9 The Soap molecule has a a Hydrophilic head and hydrophobic tail b Hydrophobic head and hydrophilic tail c Hydrophobic head and hydrophobic tail d Hydrophilic head and hydrophilic tail
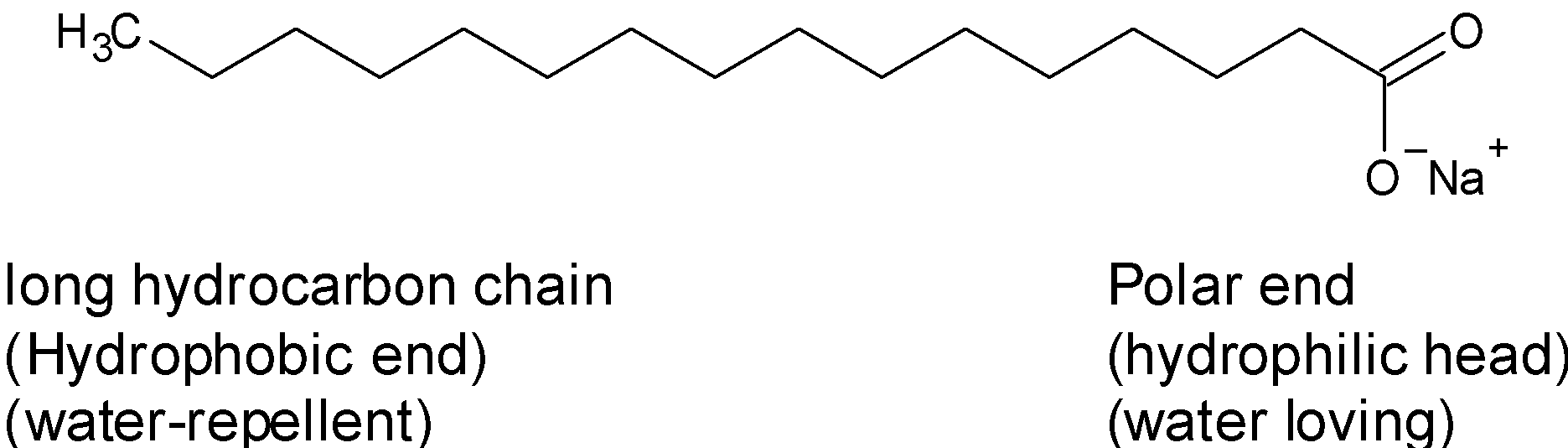
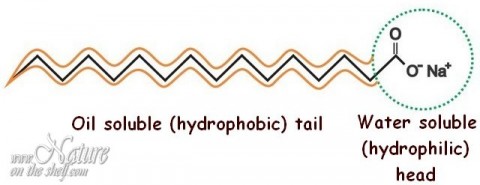
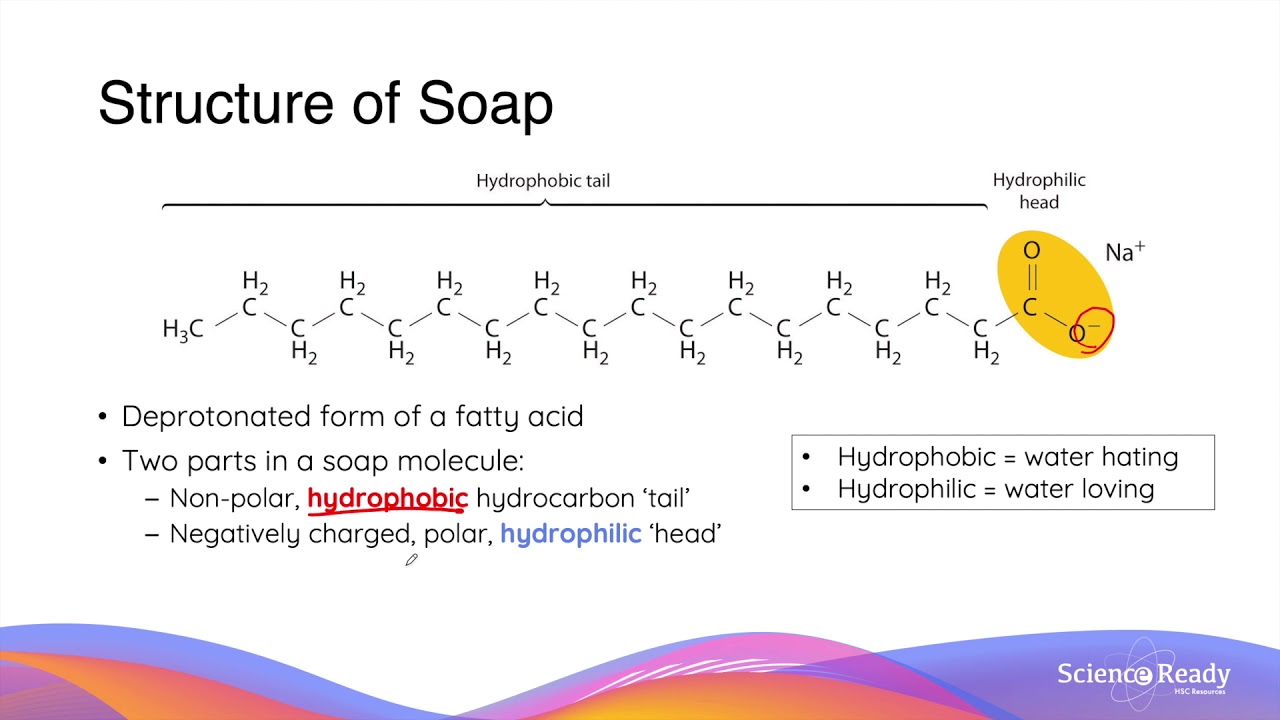

![MCQ] In the soap micelles (a) the ionic end of soap is on the surface MCQ] In the soap micelles (a) the ionic end of soap is on the surface](https://d1avenlh0i1xmr.cloudfront.net/aed2074f-aafe-447a-b1f0-adb685bf01d5/structure-of-a-soap-micelle---teachoo.jpg)