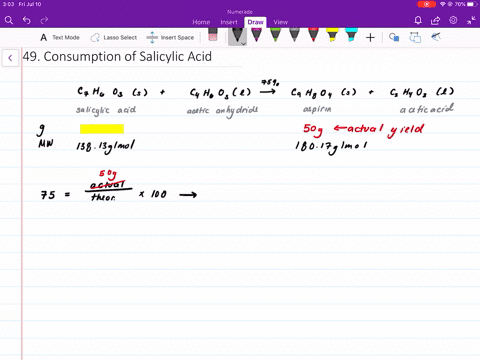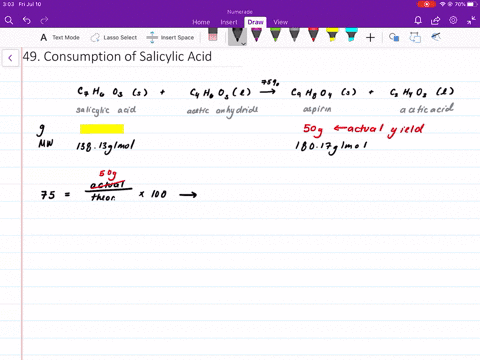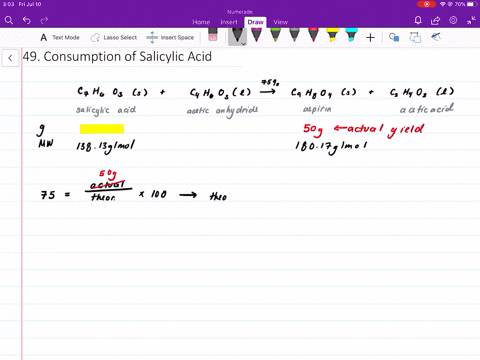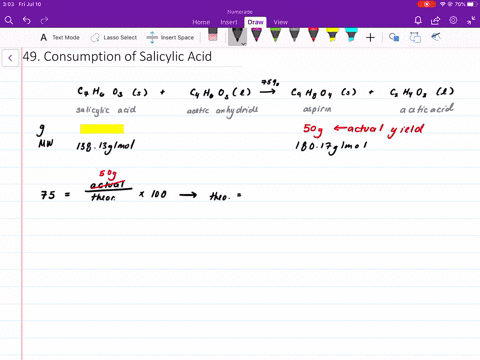
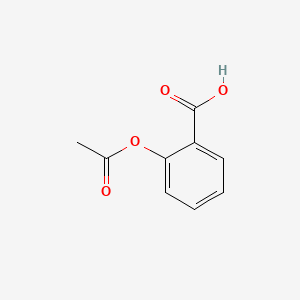
![The active ingredient in aspirin is acetyl salicylic acid with\\[{{K}_{a}}=\\text{ }4.0\\times {{10}^{-9}}\\] . The pH of the solution obtained by dissolving two aspirin tablets (contain 0.36 g of acetyl salicylic acid in The active ingredient in aspirin is acetyl salicylic acid with\\[{{K}_{a}}=\\text{ }4.0\\times {{10}^{-9}}\\] . The pH of the solution obtained by dissolving two aspirin tablets (contain 0.36 g of acetyl salicylic acid in](https://www.vedantu.com/question-sets/214d4d7b-aa1e-4b43-8585-31cd4abd95614088496745223917951.png)
The active ingredient in aspirin is acetyl salicylic acid with\\[{{K}_{a}}=\\text{ }4.0\\times {{10}^{-9}}\\] . The pH of the solution obtained by dissolving two aspirin tablets (contain 0.36 g of acetyl salicylic acid in

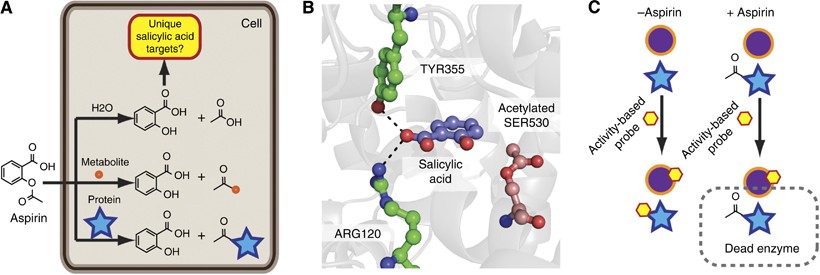
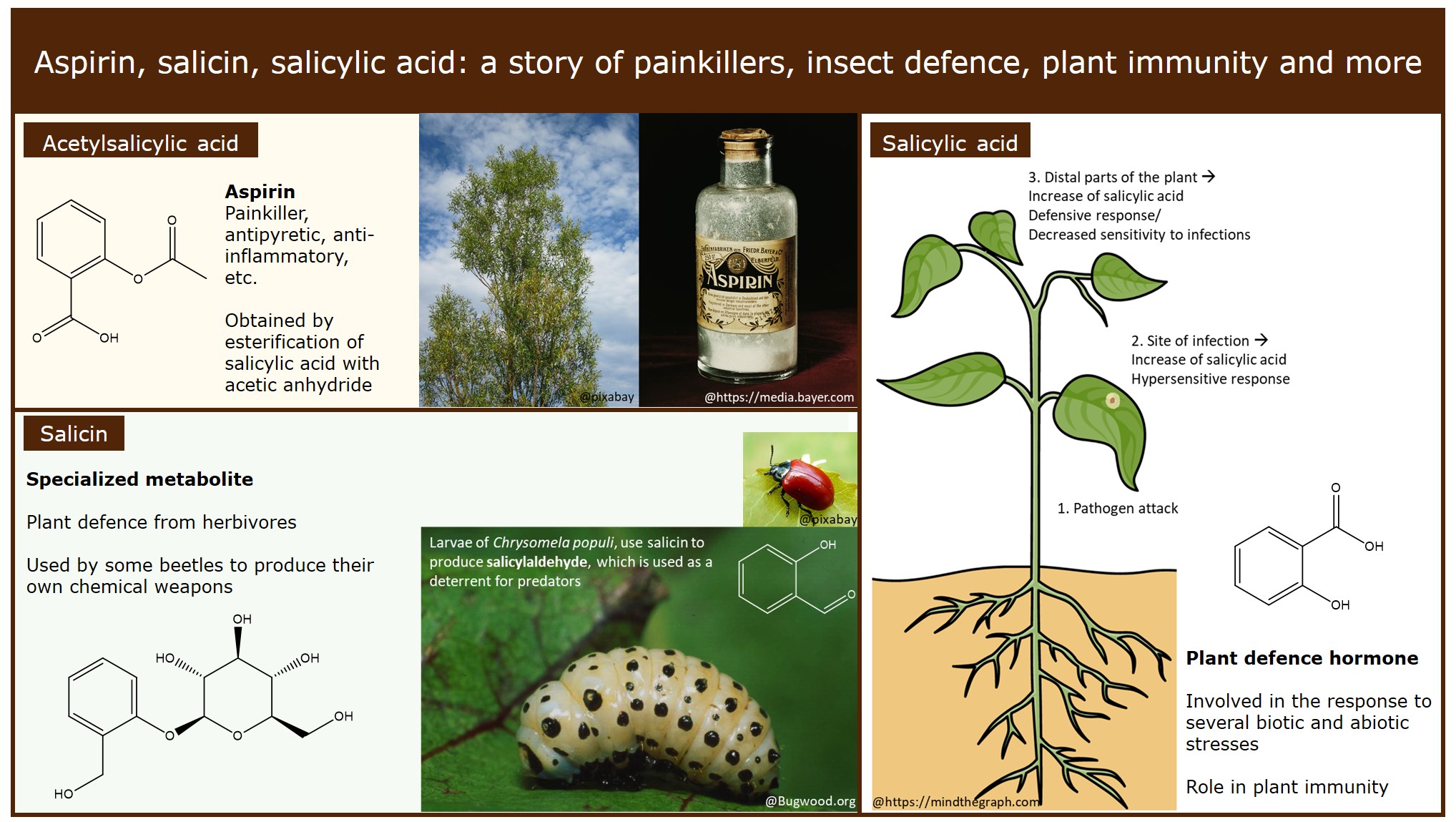
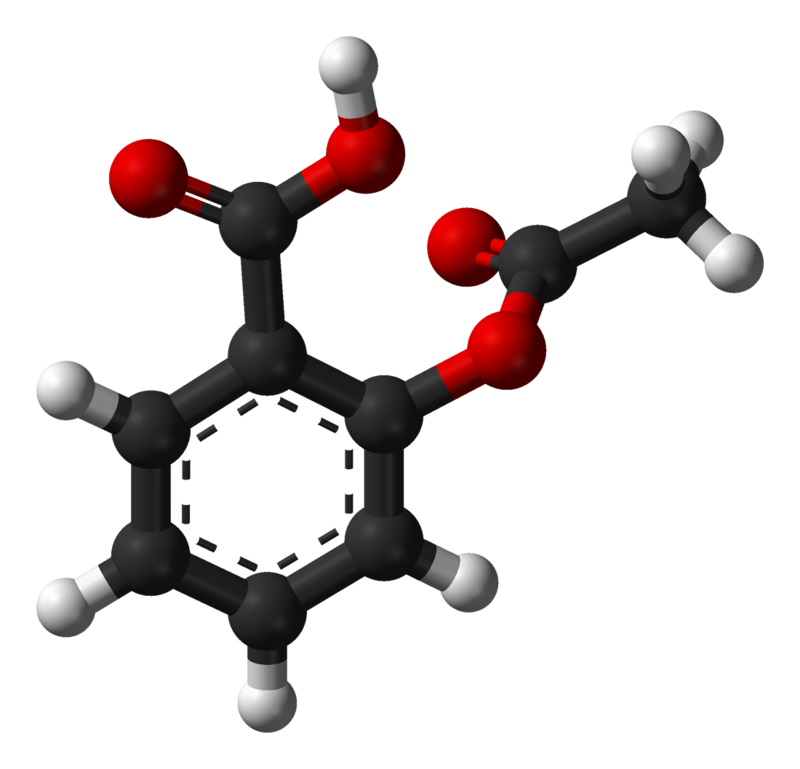
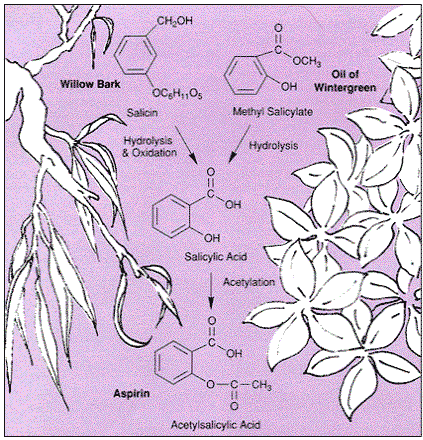
Chemical structures of salicylic acid and its synthetic and natural... | Download Scientific Diagram

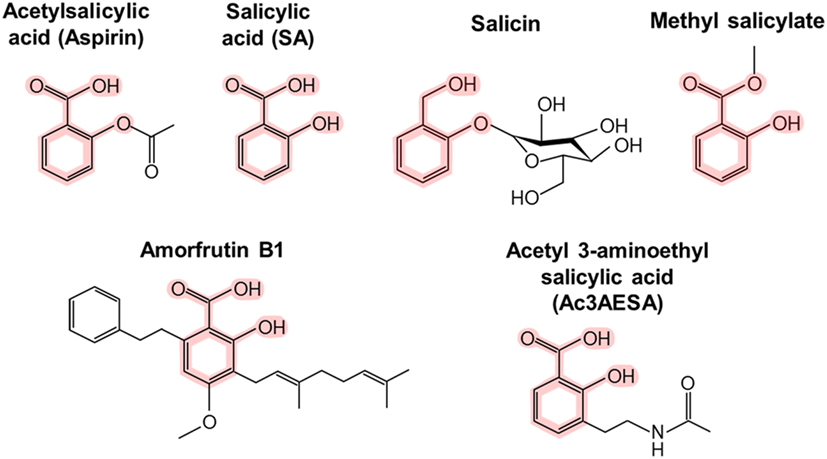
Structural representations of acetylsalicylic acid (1a) and salicylic... | Download Scientific Diagram
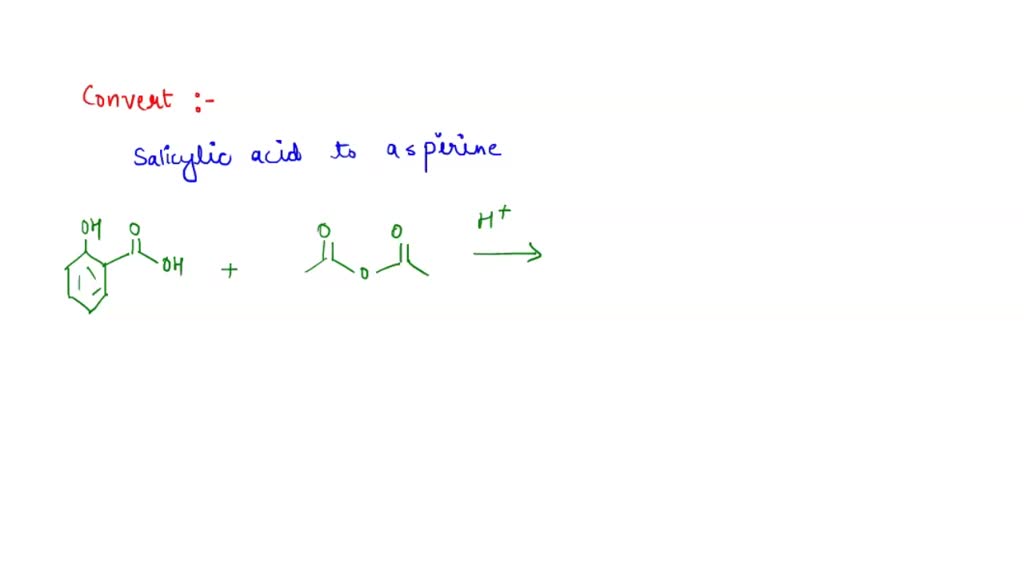
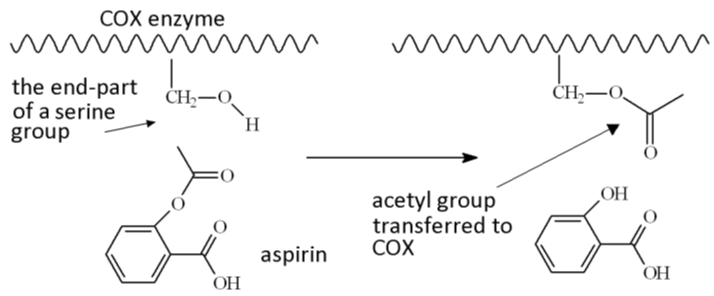
SOLVED: Carboxylic acid and phenol. Why would salicylic acid contain two acidic groups? Is it desirable to convert salicylic acid into aspirin (acetylsalicylic acid)?
When synthesizing aspirin in the lab, the product turns out to be a yellow solution and brown ppt when salicylic acid is reacted with acetic anhydride and H2SO4 (as a catalyst). What